Saudi Arabia is investing heavily in its tourism industry, shifting its focus beyond the pilgrimage sites of Mecca and Medina to its diverse natural landscapes, vibrant cities and growing list of UNESCO World Heritage Sites.
The government aims to create 1.6 million jobs in the industry and grow tourism’s share of GDP to 10 percent by 2030. When Vision 2030 was announced in 2016, the sector represented just 3 percent of GDP, and has already grown to 7 percent, which is promising.
As tourism continues to grow, both in Saudi Arabia and around the world, so too does its energy consumption, driven by a need to fuel hotels, resorts, experiences and mobility. In fact, Green House Gas impact of tourism is approximately 8-11%, of which aviation contributes some 17%. Additionally, there is a tendency for visitors to use and consume more resources than local people, especially in poorer areas of the world.
It is vital, therefore, that tourism accelerates its transition to renewable energy. By embracing solar and other clean energy sources, the industry could significantly reduce the world’s carbon footprint and help to combat climate change.
It could also attract even more tourists. The Expedia Sustainable Travel Survey showed that 90% of consumers now look for sustainable travel options and are willing to pay an average of 38% more to ensure their experiences do not negatively impact the planet. Consumer surveys in other areas tell a similar story and are powerful incentives to drive the transition.
Putting renewable energy at the heart of tourism strategies is essential, but it is often complex to deliver. Our experience at Red Sea Global has produced several learnings that provide value for any executive tasked with driving a green energy transformation and show that it is always possible to build a future driven by clean power.
A COMPELLING INVESTMENT OPPORTUNITY
Delivering renewable energy requires significant investment in technology, but even high upfront costs pale in comparison to the financial and environmental rewards to be reaped in the long term.
Across the world, the signs are positive. In the US alone, private investment in renewables hit USD 10 billion in 2022, according to Deloitte. A willingness to invest in technological infrastructure should be embedded at the very beginning of any development process and can help to attract partners and investors.
We saw firsthand the positive effect of this with a consortium led by ACWA Power bringing Foreign Direct Investment from the UK’s Standard Chartered Bank and China’s Silk Road Fund for the Utilities PPP agreement set to power The Red Sea destination with sunlight.
A similar partnership was concluded in September this year, when a multi-utilities concession agreement was signed for Amaala with EDF (Électricité de France) and Masdar, which will deliver new eco infrastructure facility will save nearly half a million tons of CO2 emissions every year.
Moreover, our historic Financial Close on the first ever Riyal-dominated Green Financing for the development of Phase One of our destination, The Red Sea. This financing arrangement was valued at SAR 14.12 billion (USD 3.76 billion).
Attracting these partnerships and support early on, ensured that from the very start clean energy would fuel both our vision and our flagship destinations, The Red Sea and Amaala, with the former set to become the world’s largest off-grid tourism destination powered by renewable energy.
Across our destinations, everything will be powered by clean energy, even the electric vehicles within our smart mobility network covering land and sea, with ambitions to power our seaplanes with electricity too. At The Red Sea, our battery storage facility – the world’s largest – will store up to 1,200MWh of power and enable us to operate fully off-grid, powered by sunlight day and night.
Our journey is just starting, but we are proud of having built renewable power into our development from the ground up. That doesn’t mean that other businesses must start from scratch. What matters isn’t necessarily the size of the investment, but the ambition, creativity, and plan behind it. Drawing on the natural attributes of a business’ location represents a huge step towards sustainability.
For a destination spread across the sun-soaked dunes and coastline of Saudi Arabia, that means investing in solar power. We have installed 760,000 photovoltaic panels across five solar farms to power The Red Sea renewably.
CROSS-INDUSTRY PARTNERSHIPS
Finding the right solution for every unique development could also involve working with expert partners. This is true of developments across all industries, but again the tourism sector shows one way forward.
For example, travel associations across Africa have partnered with a range of international universities, including the University of Brighton in the UK and the University of Ghana, as well as Africa Tourism Partners in South Africa, to encourage youth involvement in sustainable tourism initiatives. This has led to the launch of a new app in South Africa that takes users on digital tours and only displays ‘green’ businesses: encouraging knowledge-sharing, innovation, and entrepreneurship on renewable energy specifically.
Sharing best practices and requiring green standards from partners and suppliers is also vital. At Red Sea Global, cross-industry collaboration is fundamental to our vision of sustainable and responsible development. We select partners with like-minded values and the brands we have brought on board share our ambition to do better by people and planet.
RENEWING TO REGENERATE
A commitment to renewable energy should be at the heart of any business strategy and its operations. Building the infrastructure from the start is ideal, but the next best thing is starting from where you are, and making steady, incremental changes.
By investing in technology and partnering with like-minded organizations, companies can maximize their business potential without harming the planet. They can also use their position to catalyze and inspire their customers to widen the impact even more.
This, in turn, will help revive the fortunes of the natural and human ecosystems in these areas, maintaining and even improving them for future generations to enjoy.
After super-powerful chatbots such as ChatGPT-4 started becoming widely available this year, school administrators around the world moved to ban the technology from classroom education. Nearly half a dozen US districts blocked access to AI and other multimodal large language models (MLLMs) on school devices and networks, and some Australian schools turned to pen-and-paper exams after students were caught using chatbots to write essays.
Teacher resistance reached its peak when ChatGPT-4 was released in March 2023. Developed by San Francisco-based OpenAI, this generative AI can write poetry and songs, and it passed the US bar exam in the 90th percentile. MLLMs can process images as well as text, and they answer queries by looking for patterns in online data.
When asked why Seattle schools had moved to restrict ChatGPT-4 from district-owned devices, a spokesperson for the district, Tim Robinson responded: “Generative AI makes it possible to produce non-original work, and the school district requires original work and thought from students.”
However, confronted with AI’s seemingly inevitable growth, many schools are now reversing course, albeit carefully. “There’s still a fear that students will use large language models as shortcuts instead of practicing to become better writers,” says Tamara Tate, a project scientist at the University of California, Irvine’s Digital Learning Lab. She adds that if AI is here to stay then students might be better served by educational strategies that promote creative uses of the technology. “These tools can provide students with in-the-moment learning partners on a huge range of topics.”
In the view of Tate and other experts, MLLMs have several positive educational roles to play, including encouraging students to evaluate answers rather than automatically accepting them. Careful thought is needed to ensure that these potential upsides are realized, however, and to mitigate any potential downsides. How might AI-assisted education unfold?
Classroom gains and losses
Proponents of the educational uses of generative AI point to several advantages. For one thing, ChatGPT-4 has an extraordinary command of proper sentence structure, which Tate says could be especially useful for non-native speakers seeking insight into how to correctly incorporate words and phrases in real-world settings.
Xiaoming Zhai, a visiting professor who studies applications for machine learning in science education at the University of Georgia in Athens, believes that teachers also stand to benefit from using models like ChatGPT as teaching aids. The models can generate personalized lesson plans and other resources geared to the needs of individual students while assisting with grading and other mundane tasks. In Zhai’s view, that capability frees time so that teachers can provide students with more one-on-one feedback. By efficiently automating basic tasks like searching out relevant literature and materials and summarizing content, the models allow students and teachers alike to “focus more on creative thinking”.
Creative thinking will help people get the most from MLLMs. “Large language models are like search engines: garbage in, garbage out,” Tate wrote in a recent preprint paper.
Teachers can help their students develop expert prompting and search optimization strategies to generate the most helpful content. “To use the technology effectively, students need to double down on the work of revision,” Tate says. “ChatGPT-4 can generate a fluent first-draft response, but not a lot of deep content. The responses can be vague and often wrong.”
While researching this article, we asked ChatGPT-4 to tell us, in its own words, why it would be a helpful tool for education. Seconds later, the model provided a detailed answer in which it claimed it had access to vast amounts of knowledge and could respond instantly to questions in multiple languages at any time. But the model was also candid about its limitations, pointing out that if ChatGPT-4 doesn’t understand the nuances of a particular question, then it might deliver incomplete or erroneous information that could be problematic for students who rely solely on the model for answers.
Given that MLLMs may fail to support their claims with reasons or evidence, this gives teachers the opportunity to demonstrate the need for critical reasoning. “Students need to think about who said what and why in a given response,” Tate says.
Lea Bishop, a law professor at Indiana University’s Robert H. McKinney School of Law in Indianapolis, agrees that potential inaccuracies will require students to scrutinize the model’s output. “You have to develop the habit of questioning everything you see,” she says. “That means asking probing follow-up questions and triangulating with other sources of knowledge to see what matches up. I need you to show me that you’re better than the computer.”
Dealing with cheating and secrecy
Some experts worry that, for less motivated students, these sorts of models provide a tempting source of ready-made content that diminishes critical thinking skills. The predecessors to ChatGPT-4 proved themselves capable of generating essays and responses to short-answer and multiple choice exam questions. “We already have a lot of problems with students who feel that learning equates to searching, copying and pasting,” says Paulo Blikstein, an associate professor of communications, media, and learning technologies at Columbia University, in New York. “With AI, we have an even greater risk that some will take the shortest and easiest path, and incorporate those heuristics and methods as a default mode.”
Teachers can try to flag AI-generated content with software packages called output detectors. But these packages have questionable reliability, and in July 2023, OpenAI discontinued its own output detector citing concerns over low accuracy. Experts worry that models like ChatGPT-4 will increasingly put teachers into the unwanted role of having to police students who break rules on AI-generated content.
Such concerns are valid, and contributed to the initial negative responses. Blikstein says early school restrictions may be seen as a “knee-jerk reaction against something that is still very hard to understand”.
And although these bans are gradually being lifted, ChatGPT is not yet in the clear: its workings remain opaque, even to the experts. Between its inputs and outputs are billions of ‘black-box’ computations. ChatGPT is said to be OpenAI’s most secretive release yet. The company hasn’t disclosed anything about how the model was trained, and proprietary systems developed by competing companies are now driving an AI ‘arms race’ — advancing at mind-boggling speed.
Defining core skills
Does the rise of MLLMs mean writing itself will go the way of older skills, in much the same way that basic mathematical competence was rendered nearly obsolete by calculators? Experts offer a range of opinions. Taking a bullish stance, Bishop argues that functional writing skills such as spelling, grammar, and knowledge of how to organize a standard essay “will be totally obsolete two years from now”. Others see need for caution. “Without practice writing their own content, it will be hard for students to predict where and how writing mistakes are made — and then spot them in AI-generated content,” Tate says.
In Blikstein’s view, this grey area underscores a need to proceed slowly. “The stakes are high with language,” he says, adding that generative AI can be a powerful partner for enhancing — not replacing — a student’s cognition. But important questions remain. “For instance, we don’t have a good model for authorship in the area of AI-generated content,” he says. “The text appears out of the ether, and we have no idea where it came from.” For accomplished professionals, using AI to boost writing skills may not pose much of a problem. “But that’s not true for younger people who don’t understand the craft of writing to begin with,” he adds.
Blikstein also worries that AI might perpetuate educational inequities. Wealthier school districts have resources to apply the technology with an emphasis on human interaction and project-based learning, while poorer schools might move increasingly towards automation to save money. “If you settle for something cheap, it can take over your whole system,” he says. “Then five years later, it’s the new normal,” he says.
Ultimately, AI could offer an evolution in educational norms that sends educators back to basics. “We have to identify the core competencies that we want our students to have,” says Zhao. “How are we going to incorporate models like ChatGPT into the learning process? We are preparing future citizens, and if AI will be available, then we need to think about how we build competence in education so that students can be successful.”
Explore FII’s publications site for more thought-provoking articles and podcasts about artificial intelligence and the impact of technology on society.
Mobile robots can dance around a stage, perform graceful acrobatics and even lift heavy objects. But if you watched them strut their stuff for an hour or two, you would see the robots grind to a halt. Like humans, mobile robots eventually exhaust the energy that they carry, and need a recharge.
This problem is specific to mobile robots. Robots anchored to a factory floor can do heavy work all day and all night because they can draw inexhaustible energy from the electric grid. Mobility gives robots more flexibility, but at the cost of needing to recharge their energy sources — in most cases, some form of battery.Part of Nature Outlook: Robotics and artificial intelligence
The compact nature of smartphones can fool us into thinking batteries are featherweight objects. That illusion arises because modern electronics need only a trickle of energy to send signals or process data. But transporting robots or people around, or lifting a heavy load, takes much more energy. If you pick up a cordless tool, you will feel that it outweighs a corded one. An electric car that can travel for five hours (around 500 kilometres, the distance from Paris to Amsterdam) at motorway speed between recharges needs batteries that account for one-third or more of the total vehicle weight.
Mobile robots on legs, however, can’t tolerate such massive batteries. Boston Dynamics, a robotics company in Waltham, Massachusetts, sells a four-legged dog-size robot called Spot that weighs about 32 kg — one-eighth of which is batteries. But, the company states it has a typical run-time of only 90 minutes. Humanoid robots that have been developed to walk with heavy loads have the same limitations. Atlas, the company’s 1.5 metre, 89-kilogram humanoid demonstrator with two arms and two legs, can do gymnastics and lift heavy objects. But the company does not say how long it can run before it needs a recharge. For mobile robots to be more capable workers, their batteries will need greater energy density — that is, they will need to pack more watt-hours of energy into fewer kilograms of mass. “Energy density is still quite far from the power we need for robotics,” says Ravinder Dahiya, an electrical engineer specializing in robotics at Northeastern University in Boston, Massachusetts.
How serious the energy-density problem is depends on the robot’s size and structure, its function and how much energy it needs. Robots that walk can navigate stairs, the interiors of buildings and rough terrain better than wheels — but they can’t carry as big of a battery pack. Sustained flight requires even more energy, making battery weight a serious limit for anything much bigger than insect size.
The limits of lithium
Batteries have come a long way since the Italian physicist Alessandro Volta invented the earliest version of this technology in 1800. Today, the state-of-the-art power source is the lithium-ion battery, invented in the 1970s by chemist Stanley Whittingham, and now widely used in phones, laptops, tools and electric vehicles. The technology earned Whittingham, now at the State University of New York at Binghamton, a share of the 2019 Nobel Prize in Chemistry.
Batteries don’t generate energy; they store energy produced by chemical reactions that yield positive ions and electrons. The ions accumulate at one end of the battery, called the cathode, and the electrons at the other end, called the anode. The ions and electrons sit on these two ends until they are connected by a conductor, which completes the circuit and allows the electrons to flow as a current from the anode to deliver electrical power to an externally connected load — such as an electric motor — and then to the cathode. When the chemicals are used up, the battery must either be replaced or recharged by passing a current through it in the opposite direction, to reverse the reaction.

A major advantage of batteries is that they directly deliver energy in the form of electricity, whereas fossil fuels have to be burned to generate heat that drives an electrical generator. This avoids carbon emissions on the spot, although total emissions depend on how the original energy was produced. However, robots must carry the energy they use, and batteries weigh more and occupy more space than fossil fuels. An electric car, for example, needs a battery pack much larger and heavier than a fuel tank.
When lithium-ion batteries reached the market in 1991, they provided 80 watt-hours of electrical energy per kilogram of battery weight1. That meant it took a one kilogram battery to power a (then standard) 60-watt incandescent bulb for one hour and 20 minutes, making it the best battery available. Now, typical commercial lithium-ion batteries carry three times more energy per kilogram. But even such energy-packed batteries are too hefty for a walking robot to lug around.
Chemistry quest
Lithium-ion batteries are running out of steam. The chemistry has “less and less room for improvement”, says Richard Schmuch, a chemist at the Fraunhofer Research Institution for Battery Cell Production in Münster, Germany. Lithium itself is rare and expensive. The same is true for cobalt, another crucial element which can make up to 20% of the weight of the cathode in lithium-ion batteries for electric vehicles. Extracting both elements requires large amounts of energy and water. Moreover, the mining of cobalt has been linked to the exploitation of workers.
Another concern is optimizing batteries to meet the needs of robotics. “The lithium-ion battery is quite versatile,” says Schmuch. “You can adjust it for different types of operating condition,” from smartphones to cars to robots. Yet it can’t do everything cost-effectively and well. He expects new types of battery will be needed to serve the emerging demands of robotics as well as other applications.
After more than 30 years of development, lithium-ion batteries are considered to be a mature technology. Still, efforts continue to improve these complex electrochemical systems. They are assembled in units called cells, which are packaged together to provide a desired electrical output. Each cell contains — in addition to the anode and cathode — an electrolyte through which ions can move, a separator to prevent short circuits and electrical terminals that connect to other cells in the packaged battery. Extensive research has gone into the composition of each part to achieve high energy density, charging and discharging rates, reliability and longevity. Among the important successes of this technology are batteries that can be recharged as many as 6,000 times.
One attempt to enhance the performance of lithium entails making the anode and cathode from nanostructured sulfur-graphite composites rather than from standard graphite. Such lithium–sulfur batteries offer the potential of lower costs and higher energy density. These batteries have yet to be commercialized successfully, however; their use might be limited to specialized applications, such as aviation, for which minimizing battery weight is crucial to get off the ground. Battery features could be tailored by adjusting design details, such as the type of nanostructure used and how the ions and electrons flow through the battery. But what many developers want is new battery chemistries designed to meet a variety of needs (see ‘Packing in the power’).
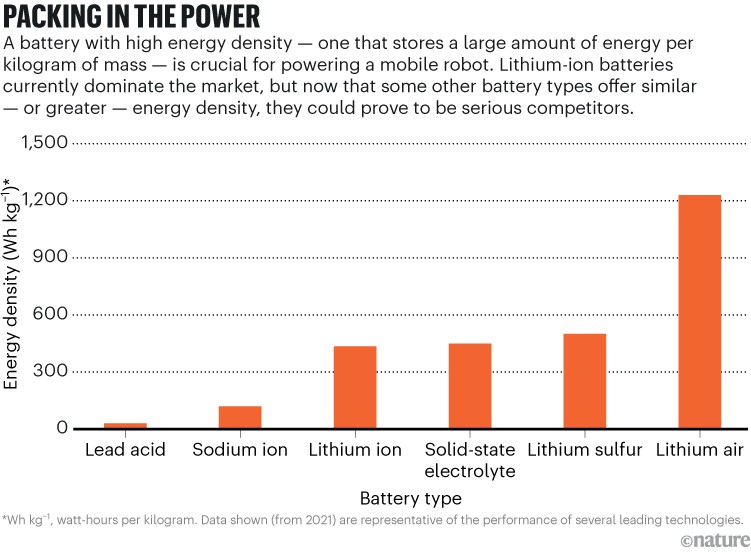
That might entail stepping back from lithium’s biggest attraction — it is the lightest metal among the elements, with an atomic weight of seven. Yet, although lithium is absolutely essential to the battery’s energy storage and release, other materials make up more of the battery’s mass. Including the packaging, only about 1% of the weight of a lithium-ion battery is lithium (most of that in the cathode). The cathode also contains more of four other metals: cobalt, nickel, aluminium and manganese. Several problems with lithium and cobalt have led to serious interest in sodium-ion batteries.
Like lithium, sodium is an alkali metal, and the chemistry of the two is so similar that researchers have pursued sodium-ion batteries as a way around the problems with lithium. One important advantage of the sodium-ion design is the ready availability of sodium in seawater and salt deposits — avoiding the supply-chain problems arising from the cost and scarcity of lithium. Sodium-ion batteries are a bit heavier per kilowatt-hour of energy — sodium’s atomic weight is 23, more than triple that of lithium. Still, lower material costs are expected to make sodium-ion batteries significantly cheaper. An even bigger benefit of switching to sodium would come from reducing or eliminating the need for cobalt in the cathode, which has been demonstrated in several samples.
“Sodium ions are definitely gaining traction,” says Schmuch, citing development of them in Germany, where Fraunhofer is working with industry, and in China, where Contemporary Amperex Technology (CATL) in Ningde — the world’s leading manufacturer of lithium-ion batteries for electric vehicles — rolled out the first generation of its sodium-ion battery in 2021. This April, Chery Automobile in Wuhu, China, announced plans to install CATL sodium-ion batteries in its cars. Also in April, CATL said it had developed a new electric vehicle battery with an energy density of 500 watt-hours per kilogram. This battery employs a different technology, which CATL have not identified.
Solid-state solutions
Another way to change battery chemistry is to change the state of the electrolytes, replacing the conductive liquids used in lithium-ion batteries with conductive solids. Advocates think such solid-state batteries offer the best prospects for preventing the potentially deadly fires seen with lithium batteries, as well as for improving energy density and reducing costs.
The fire hazard comes from filamentary deposits of metallic lithium called dendrites that grow in electrolytes in the batteries. Lithium atoms present in the solvent crystallize to form metal filaments that spread like plant roots. The metallic lithium is conductive, and as the dendrites spread they can short circuit the battery and ignite fires. Sodium is much less prone to dendrite formation, and developers think that this quality makes sodium-ion batteries significantly safer than lithium-ion batteries. They would also be lower cost and in the long term potentially offer higher energy density.
A wide range of solid-state batteries are in development. Lithium is still a popular material because of its light weight, high energy density and rechargeability. But some researchers are exploring other metals with the hope of avoiding the known problems of lithium.

Switching one type of lithium battery in particular from liquid to solid-state electrolytes has led to a big advance in efficiency. This beneficiary is the lithium-air battery, which produces power from the oxidation of lithium atoms by oxygen from the air. As with sodium-ion batteries, energy density was not the main goal for many people working on solid-state batteries. “The reason for developing a solid-state electrolyte was to make the lithium-air battery more safe and to make recharging cycles more stable,” says Mohammad Asadi, a chemical engineer at the Illinois Institute of Technology in Chicago.
However, when Asadi and his colleagues from Argonne National Laboratory in Lemont, Illinois, built an experimental solid-state lithium-air battery2, they were surprised to discover that the technology brought significant benefits in energy density as well. The device transferred four electrons per reaction rather than one or two electrons per reaction that lithium-air batteries normally produce. As a result, Asadi says, the solid electrolyte “helps us store three to four times more energy per unit weight” than is possible with conventional lithium-ion batteries.
In fact, their new solid electrolyte changed the chemistry between oxygen and lithium. In standard lithium-air batteries, oxygen molecules from the air react with lithium atoms to produce one of two compounds. The reaction between one lithium atom and one oxygen molecule produces lithium superoxide (LiO2), which yields one electron. The reaction between two lithium atoms and one oxygen molecule produces lithium peroxide (Li2O2), which yields two electrons.
Asadi’s team made its solid electrolyte by combining nanoparticles containing lithium, germanium, phosphorous and sulfur (Li10GeP2S12) with a polymer. In this structure, two lithium atoms can combine with a single oxygen atom to yield lithium oxide (Li2O) and four electrons. That’s hard to do because it requires splitting an oxygen molecule (O2) to produce a single oxygen atom. The test cell, only the size of a coin, was a proof of concept. According to Asadi, this prototype shows that it will be possible to attain a specific energy of one kilowatt-hour per kilogram — higher than is possible with today’s lithium-ion technology.
Power-studded structures
Increasing the energy density of batteries makes it possible for these power packs to weigh less, which in turn would allow mobile robots of a given size to do more work. Battery design for such robots involves much more than chemistry. And one way to realize this would be to have smaller batteries serve as structural elements of a robot — not just storing energy but also becoming parts of its torso and legs to help it walk and balance. The idea comes from biology. Our bones are not just structural support — they also contain bone marrow that produces blood cells. “Multifunctionality is critical” when you’re building robots that move around, says Nicholas Kotov, a chemical engineer at the University of Michigan in Ann Arbor.
“Robots are biomimetic, and the smaller the robot, the more biological concepts would need to be there,” Kotov says. He and other roboticists call two-legged robots humanoid because they walk upright and have similar body mechanics to people. “We want to keep robots light and consuming as small as an amount of energy as possible,” Kotov says. “If a battery just sits there and does nothing else [but provide power], it is not enough.”
Kotov’s group is particularly interested in drones, in which, he says “every gram counts, and if the battery can serve multiple functions, we can have more functional space”. His team is now working on structural batteries for military drones, although not much of that work has been disclosed yet. Military laboratories have also worked on humanoid robots for missions such as working inside radiation zones and checking for insurgents hiding inside buildings in combat zones.
Some materials used for battery energy storage are particularly well-suited for also being structural elements. For example, Kotov says, “zinc is a very good case for structural batteries”. It is inexpensive, stores energy well and the metal is stable in air. His lab demonstrated a biomorphic zinc battery that could store 72 times more energy than a lithium battery of the same volume3. However, trade-offs are
inevitable. Zinc batteries have limited rechargeability, so they would be best kept stashed away for infrequent use.Another promising multifunctional material is aluminium, which, last year, showed rapid rechargeability over hundreds of cycles at temperatures up to just above the boiling point of water — and without forming aluminium dendrites4. The researchers project a cost of less than one-sixth of that of comparable lithium-ion batteries with a similar energy capacity.
Kotov is also developing aramid fibres to provide structural strength for battery casings and internal battery structures3. These fibres have a fortunate combination of features including strength, flexibility and hardness that makes them useful for protective shielding. One particularly helpful attribute is their ability to block dendrites from growing between the electrodes. Moreover, aramid offers an environmental advantage — it can be made from recycled Kevlar, a strong, lightweight material, and when the batteries are worn out, the fibres can be recycled for further uses.
Energy beyond batteries
By 2030, Dahiya expects the development of energy sources for mobile robotics to broaden well beyond batteries. Some of these concepts have their roots in biology.
One example is equipping robots that operate at remote sites with energy harvesters, to collect energy from their local environment to top up their stored energy5. Robots can collect energy in the form of radio waves or sunlight, or from a thermal gradient. However, energy harvesting is not as efficient as heat pumps or wireless chargers, but it can operate in any suitable environment without special charging equipment. And there have been demonstrations of tiny bacterial-driven microbial fuel cells, or ‘biobatteries’, that could harvest material from the local biota to provide supplemental power6.

Another concept borrowed from biology is distributing energy in various ways through the robot’s body rather than concentrating it in a single battery backpack as used on some experimental humanoid robots5. Humans have three types of energy storage: triglycerides in fat cells, glycogen clusters around muscles and ATP that’s produced by mitochondria. Those systems evolved to serve different energy needs. “Humans and animals require fast energy and slow energy,” says Kotov. They need the fast energy to sprint, as well as slow energy to walk for many kilometres.
Robots and drones likewise have different needs at different times. A humanoid robot needs fast energy to lift a heavy load or run up stairs, and slower energy to patrol a field or a car park. Batteries are fine for a steady walk or jog, but not for a sprint. This gap has led to an interest in equipping robots with a different type of device — a supercapacitor — that delivers electrical energy much faster. Instead of using chemistry to store energy, a supercapacitor stores an electrical charge that it collects over a period of time from an electrical circuit. When a burst of energy is needed, the system discharges the stored electrons extremely quickly. Supercapacitors are used in regenerative braking systems in vehicles and can withstand many more charge–discharge cycles than can batteries7. In the future, they could give mobile robots a quick start — or a quick stop.
The lithium-ion generation of batteries put smartphones in our pockets and electric cars on our roads. Researchers are now in the early stages of developing a generation of portable energy sources that will be lighter, more efficient and potentially cheaper.
Yet costs might still be troublesome, cautions materials chemist Donald Sadoway at the Massachusetts Institute of Technology in Cambridge. Sadoway, who focuses on energy-storage technologies, sees little interest in “new battery chemistries whose price-to-performance ratio is less favourable than that of today’s lithium-ion”. It is unclear, he says, whether “the commercial opportunity is large enough to attract the investment needed for the requisite research and development to invent the new technology and to bring it to market”.
The prospects look bright to material scientist Shirley Meng, who is chief scientist at the Argonne Collaborative Center for Energy Storage Science at Argonne National Laboratory. She says using the oxygen from the air as a cathode is “the ultimate dream of battery scientists” as it offers high energy with light weight. “Good progress has been made” on the lithium-air battery, on which Argonne collaborated, but she says “we still face a lot of challenges in understanding and overcoming the limiting factors in enabling air cathodes”.
Meng predicts that the sodium battery, free of elements such as lithium, nickel and cobalt, “will find its niche to shine” because of its high ratio of performance to cost. Solid-state batteries, “offer the possibility of achieving the highest volumetric energy density in robotic applications [in which] space is limited”, she says. They also offer unique flexibility in packaging and can operate at extreme temperatures, which is important for some special-purpose robots. Meng is optimistic that developers “will offer a wide variety of battery solutions to different types of robot application”, with the potential “to unlock applications that were previously not possible”.
doi: https://doi.org/10.1038/d41586-023-02170-y
This article is part of Nature Outlook: Robotics and artificial intelligence, an editorially independent supplement produced with the financial support of third parties. About this content.
References
- Duffner, F., Kronemeyer, N., Tübke, J., Leker, J., Winter, M. & Schmuch, R. Nature Energy 6, 123–134 (2021).Article Google Scholar
- Kondori, A. et al. Science 379, 499–505 (2023).Article PubMed Google Scholar
- Wang, M. et al. Sci. Robot. 5, eaba1912 (2020).Article PubMed Google Scholar
- Pang, Q. et al. Nature 608, 704–711 (2022).Article PubMed Google Scholar
- Mukherjee, R., Ganguly, P. & Dahiya, R. Adv. Intell. Syst. 5, 2100036 (2023).Article Google Scholar
- Gao, Y., Mohammadifar, M. & Choi, S. Adv. Mater. Technol. 4, 1900079 (2019).Article Google Scholar
- Partridge, J. & Ibrahim Abouelamaimen, D. Energies 12, 2683 (2019).
FII Institute’s partnership with HEC Paris is one that thrives on the hopes of furthering one agenda: making a positive impact on humanity.
FII Institute’s THINK Pillar, which is guided by experts, partnered with esteemed academia, and run through a passionate team, aims to empower the world’s brightest minds to identify the solutions that make a difference in lives globally.
THINK achieves this by collaborating with high-caliber partners, such the reputable HEC Paris, to build momentum towards change and pursuing thought leadership in the service of humanity.
To that extent, Daniel Halbheer, an Associate Professor of Marketing and the Academic Director of the Climate & Earth Centre at the HEC Paris S&O Institute at HEC Paris was chosen as the holder of the FII Institute research chair on “Business Models for the Circular Economy”.
Business leaders, consumers, and governments alike are increasingly becoming aware of the enormous resource and waste footprints resulting from the “take-make-dispose” approach of the linear economy. The resource and waste footprints are key drivers of the world’s most pressing environmental problems, including global warming, biodiversity loss, and pollution.
To tackle these problems, firms must develop innovations across product design, recovery of end-of-life products, and recycling technologies that enable the transition from a linear to a circular economy. In addition to closing the loop, firms must also find new ways of extending the loop, for example through product reuse, repair, and remanufacturing.
The purpose of this research chair on Circular Economy is thus to develop new business models that drastically reduce resource and environmental footprints and at the same time have economic and social benefits.
This report covers Halbheer’s achievements for the Chair during 2022—a year in which a lot of effort was put into research, teaching, and outreach activities.
Research
- The paper “Carbon Footprinting and Pricing Under Climate Concerns” (joint with Marco Bertini, Stefan Buehler, and Don Lehmann) was published in the Journal of Marketing. For this paper, Halbheer won the 2021 Rigor & Relevance Research Award by the Swiss Academy of Marketing Science.
- The paper “Managing Service Shutdowns: Cash Refunds or Vouchers?” (joint with Rachel Chen, Eitan Gerstner, and Paolo Roma) has been accepted for publication in the International Journal of Research in Marketing.
- The manuscript “Product Design and Consumer Participation in Circular Business Models” (joint with Stefan Buehler and Rachel Chen) has been published on the Social Science Research Network repository. This paper was presented in a research seminar at Bocconi University, and it has also been submitted to several research conference scheduled in 2023.
Teaching
- The newly created “Climate and Business Certificate” (joint with Igor Shishlov) took place between May 2 and June 1, 2022. Because of this course, Halbheer and Shishlov and were awarded a highly selective CAD 10,000 Case Study Grant by the Monitoring and Evaluating Climate Communication and Evaluation Project to create a package for climate change education.
- The newly launched “HEC Climate Days” (joint with François Collin and Igor Shishlov) took place on May 30-31, 2022 as an integral part of the Climate and Business Certificate.
Outreach
- The article on “Business Schools Must Do More to Address the Climate Crisis” was published as a digital article in the Harvard Business Review.
- The article on “Business School Collaboration: Embracing Our Responsibility Towards Our Planet” was published in the EFMD Global Focus Magazine.
- The short piece “A Circular Economy That Benefits Businesses and the Planet” was published in the BNP Paribas Perspectives.
For 2023, a number of impactful activities are underway, taking the partnership between the two entities to new heights whilst changing the world one step at a time. The Institute believes that at the intersection of human need, technological feasibility and economic viability are the answers that will shape a better world for all.



